According to industry forecast, the global UV curing coating market is expected to grow from US$6.5 billion in 2020 to US$11.4 billion in 2025 with a compound annual growth rate of 12%. UV coatings provide a high-gloss surface, are environmentally friendly, wear-resistant, fast-drying, and offer a variety of performance properties. The continuous introduction of environment regulations has driven the growing popularity of green coatings in the industry and the demands for UV curing coatings have also increased. However, during the COVID-19 pandemic, the electronic and industrial coating industries suffered from a decline in sales, affecting the demands for UV curing coatings.
Due to increasingly stringent regulations on emission reduction, UV curing coatings are more accepted in regions such as Europe and North America. However, further development is needed in the Asia-Pacific and MEA (Middle East and Africa) regions. UV curing technology can be broadly divided into UV free radicals and UV cationic systems. Featuring excellent processing performance, UV cationic coatings have been widely applied in metal cans, coil steel and flexible packaging industries, which are mainly available in European and American markets.
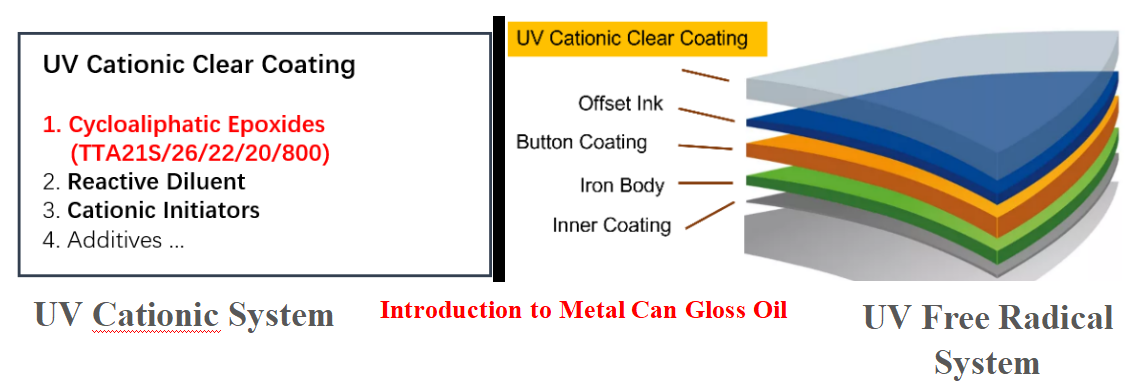
Here is a brief comparison of the two technologies in terms of the system and performance.
UV cationic and free radical materials differ greatly, but they have similar general composition. Cationic system is dominated by epoxy materials, but the reaction rate of conventional bisphenol A-type epoxy resins is slow, so cycloaliphatic epoxy resins/oxetane materials are used more widely; the commercialization of free radical system has been very mature and more solutions are available with epoxy/polyester/polyurethane modified acrylic ester materials.
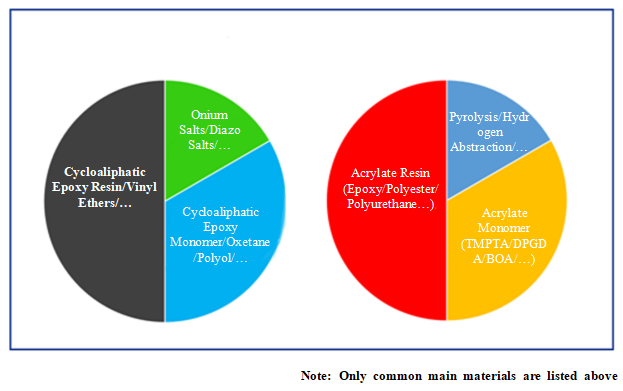
Compared with free radical system, UV cationic system has fewer options for raw materials, and the polymer mainly consists of cycloaliphatic epoxy resin with low viscosity. Taking the following cycloaliphatic epoxy resins produced by Tetra as an example:
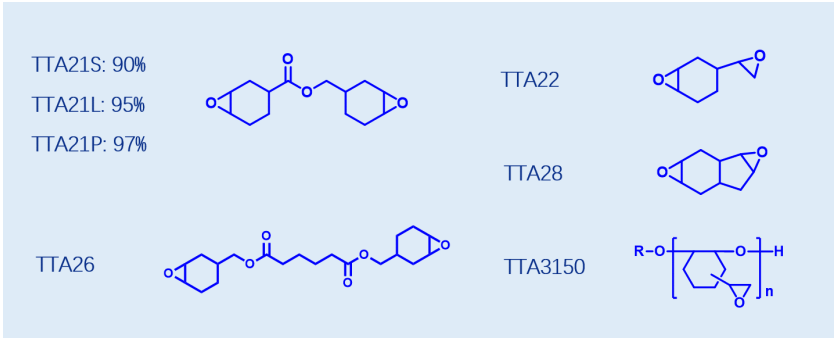
The materials mentioned above are widely applied in UV ink and coating industry, among which TTA21 with various purity specifications is the featured product. As the continuous significant increase in the application of cationic UV coatings/inks, it is expected that the use amount of cycloaliphatic epoxy resins, represented by TTA21 3 4 epoxycyclohexylmethyl 3 4 epoxycyclohexanecarboxylate, will also increase.
Tetra specialty epoxy resins are always committed to R&D and development of cycloaliphatic epoxy resin products. With complete category and sufficient productivity, the Company has made adequate product reserve for the application of various cationic UV curing scenarios.
In specific ink/coating applications, in addition to the need for UV light to provide curing energy, there are great differences in performance and reaction characteristics of both systems, as shown below:
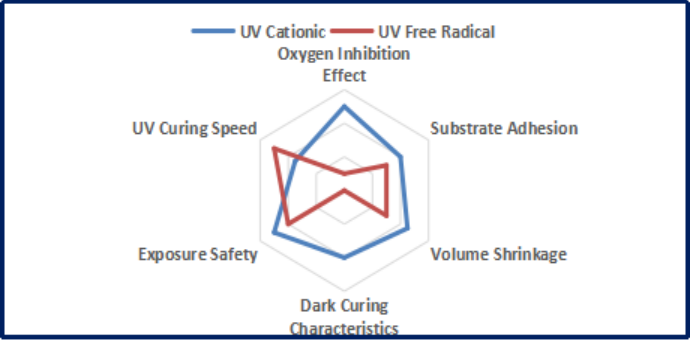
1. Oxygen Inhibition Effect
UV cationic system is not affected by oxygen inhibition, but it is sensitive to water and humidity may affect the curing efficiency of cationic system. On the contrary, UV free radical system is greatly affected by oxygen inhibition.
2. Substrate Adhesion
Generally, for difficult-to-adhere substrates such as glass/metal and high-density plastics, UV cationic system exhibits better adhesion than UV free radical system.
3. Volume Shrinkage
The shrinkage rate of UV free radical system is over 10% in general while that of UV cationic system can be controlled at 1-3%, which is an optimal solution to the volume shrinkage.
4. Dark Curing Characteristics
After the UV light source is turned off, the UV cationic system can continue to react inwardly to complete the post-curing of the material, which is well suitable for thick coating application; heating is obviously helpful to improve the cationic post-curing speed; however, UV free radical system is a reaction system that will stop chemical reaction once the UV light source is turned off.
5. Exposure Safety
The reaction degree of UV cationic system is close to 100%, and its safety can be certified with REACH/FDA testing, making it suitable for food packaging and other relevant fields.
6. UV Curing Speed
In general, the curing speed of UV free radical system is faster than that of cationic system. The cationic surface will dry faster for products that are affected by oxygen inhibition, but the actual curing speed is not as fast as free radical system. The reaction can be accelerated by heating and ultimately achieve a very good degree of completion.
UV cationic system can be blended with UV free radical system in any proportion, known as UV hybrid system, it can overcome the shortcomings such as the relative slow curing speed of UV cationic system, large shrinkage rate of UV free radical system and being influenced by oxygen inhibition. With the same film thickness, the curing energy is not significantly different between the two systems.
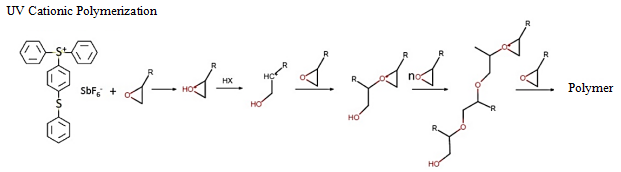
UV cationic system performs ring opening reaction by relying on the active site of Lewis acid generated by the initiator. Materials that commonly affect the initiator’s activity in the formula include organic pigments of azo compounds (protected by modification), initiators such as TOP/819/907 containing P, S and other elements and multi-stage amines similar to 115.
Humidity has a significant effect on the curing of UV cationic system, so it is advisable to control the ambient humidity below 50%. Heating can also accelerate the reaction rate.