Cycloaliphatic epoxy resins can undergo cationic photopolymerization reactions, and their UV curable adhesive has advantages such as low volume shrinkage, strong adhesion, and good weather resistance. Especially when used in free radical-cationic hybrid photopolymerization systems, they can synergize with free radical-cured products, complementing each other's strengths and weaknesses. This not only effectively improves the curing speed but also allows for use with more oligomers and reactive diluents, making it widely used in photopolymer coatings, inks, electronic adhesives, 3D printing, and other materials.
In order to provide better technical support and a better understanding of the application of cycloaliphatic epoxy resins in free radical-cationic hybrid photopolymerization systems, TATRA selected TTA-21 (3,4 epoxycyclohexylmethyl) as the cationic cured product for comparison of cured product properties with epoxy acrylates and some reactive diluents. The cationic photoinitiators used were thiol salts (TTA UV-692) and iodine salts (TTA UV-694), and the free radical photoinitiator used was 1-hydroxycyclohexyl phenyl ketone (184 photoinitiator).
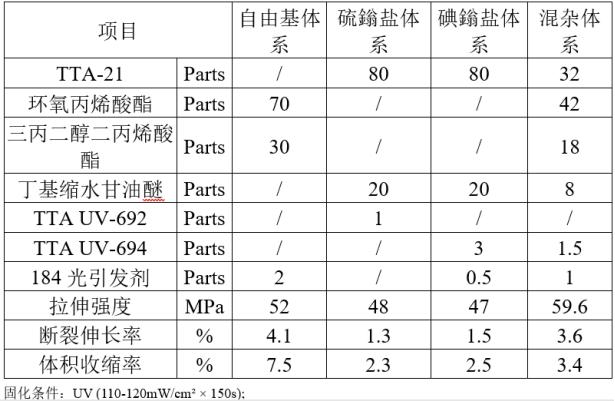
The physical properties of the cured material are as follows:
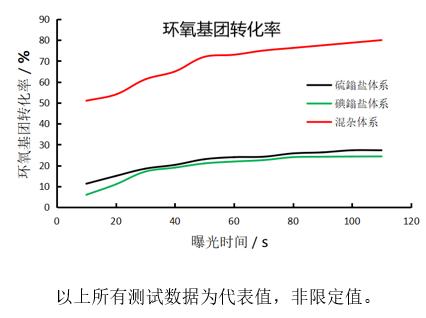
Main advantages of mixed photocuring system with cycloaliphatic epoxy resin:
Free radical photoinitiators sensitize cationic photoinitiators, increasing the utilization rate of cationic photoinitiators.
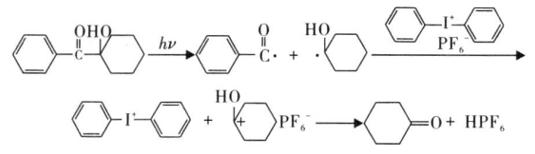
In addition, the free radical component provides more active hydrogen for the cationic component, while reducing the impact of moisture on the cationic component, thus accelerating the generation of protonic acid. The number of cationic active centers increases, and the reaction rate and degree increase as well.
The mixed system contains both free radical polymerization and cationic polymerization reactions. The free radical polymerization reaction is fast and can quickly form a rigid polymer skeleton structure under light. Subsequently, the cycloaliphatic epoxy resin continues to polymerize and crosslink on this skeleton structure basis through the cationic polymerization reaction, forming a secondary structure. The two structures are interwoven and intertwined with each other, making the structure less likely to be damaged under external forces and exhibiting high tensile strength.
When prepolymers and monomers undergo free radical polymerization, the distance between them changes from the van der Waals distance before curing to the covalent bond distance after curing, resulting in large volume shrinkage and causing problems such as large internal stress and poor adhesion. When epoxy compounds undergo cationic ring-opening polymerization, the distance between the monomer molecules changes from the van der Waals distance to the covalent bond distance after curing, leading to volume shrinkage. On the other hand, the structural unit size formed by the cationic ring-opening polymerization of the epoxy monomer is larger than the monomer molecule, resulting in volume expansion. The combination of these two effects results in a smaller volume shrinkage during the solidification of epoxy compounds in the free radical-cationic mixed photocuring system than in a standalone free radical photocuring system.
The application of cycloaliphatic epoxy resin in the mixed photocuring system provides more choices and possibilities for the research of UV curing and cationic UV coating technology and the development of special new materials with specialty epoxy resins.